Key Questions:
- What is the difference between heat and temperature?
- How does a heater warm up a cold room?
- Why can the Sun make us warm?
- Why does my cold drink become warm?
Heat Transfer
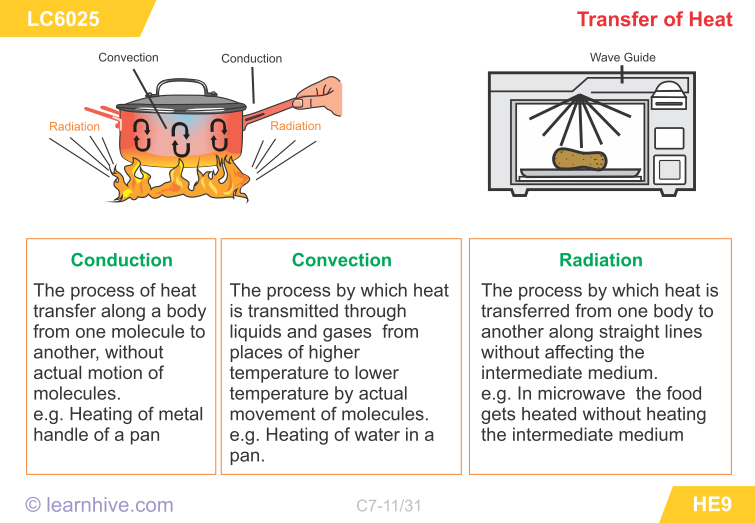
Three Types of Heat Transfer
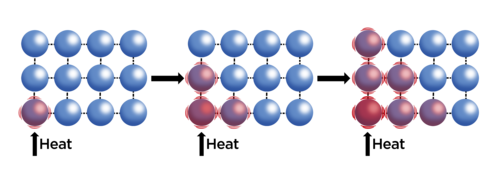
Conduction
- Occurs when the particles in an object vibrate in place but collide with neighbouring particles passing kinetic energy to them.
- žThe particles do not leave their original position.
- Conduction occurs in most solids.
Convection
- Occurs when warm fluids move from one place to another.
- Occurs in liquids and gases.
Convection Currents: When air warms, the particles gain energy, spread out, become less dense and rise. As it cools, the particles lose energy, get closer together, become more dense and sink.
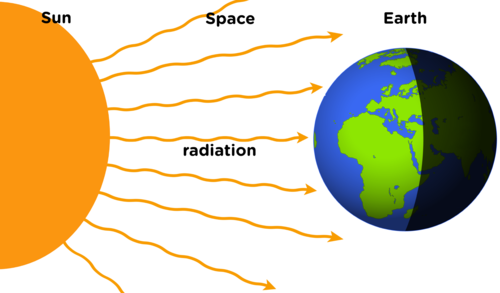
Radiation
- Occurs when electromagnetic waves carry energy from a source to another object. The object absorbs the energy of the electromagnetic wave.
- There are no particles involved.
- The waves can travel in a vacuum.
Radiant Energy can be:
1. Reflected: Shiny, smooth, light-colored surfaces.
2. Absorbed: Dull, rough, dark-colored surfaces. Object will become warmer.
3. Transmission: The radiant energy passes through. Ex. Plastic, glass, paper, etc.
1. Reflected: Shiny, smooth, light-colored surfaces.
2. Absorbed: Dull, rough, dark-colored surfaces. Object will become warmer.
3. Transmission: The radiant energy passes through. Ex. Plastic, glass, paper, etc.
Radiation travels through Electromagnetic Waves
Bill Nye: Heat
Applications of Heat Transfer: Refrigeration
Specific Heat Capacity
The amount of heat needed to raise the temperature of 1.00 g of the substance by 1.00˚C.Used to measure the amount of heat transfer.
Different materials have different capacities for storing heat. This depends on:
Different materials have different capacities for storing heat. This depends on:
- Molecular structure
- Phase
- Molecular mass
- Shape
- Surface area
Heat Capacity
While specific heat capacity tells us about how much heat a MATERIAL can store. Heat capacity tells us about how much heat an object can store.
žHeat capacity depends on two things:
ž1) the material that make up the object
ž2) How much mass the object has.
ž* Remember with specific heat capacity the mass is always set to 1 g
žHeat capacity depends on two things:
ž1) the material that make up the object
ž2) How much mass the object has.
ž* Remember with specific heat capacity the mass is always set to 1 g