Separating Mixtures
• Whenever fluids are used to help solids flow, either by simply mixing or by dissolving, the solids must later be recovered.
• You will want to know the characteristics of each of the components of the mixture and what makes them different so that you can design ways to separate one from the other. It comes down to what will attract and separate.
• This topic explores different separation methods.
• You will want to know the characteristics of each of the components of the mixture and what makes them different so that you can design ways to separate one from the other. It comes down to what will attract and separate.
• This topic explores different separation methods.
Chromatography
Desalination
•To remove salt from salty water
• Used in areas of the world that do not have access to fresh water.
• It is a process of removing water from the solution is called Dehydration
•This process uses lots of energy and is very expensive to operate as it is generally done in Hydro Plants.
• Used in areas of the world that do not have access to fresh water.
• It is a process of removing water from the solution is called Dehydration
•This process uses lots of energy and is very expensive to operate as it is generally done in Hydro Plants.
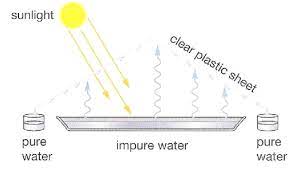
Tent Method
• Tent Method: only useful in very sunny areas. Set up an a-frame tent over a large pool of salt water. The sun hits the tent, heating the water up, causing evaporation. The evaporated molecules are pure water, and it leaves behind the salt. Note: no boiling is occurring. It is also hard to recover thee solute from this method.
- is inexpensive, but slow, and only practical in areas which receive a lot of bright sunlight
Distillation
•a separation method that allows all liquid fractions of a mixture to be separated from each other and collected in different containers.
- Because each part or phase of the solution has different boiling points, each phase can be separated at different temperatures.
- In any distillation, the mixture is heated so that at least one part begins to change into a gas (vaporize). The gas travels up and away from the mixture and the heat. In a separate chamber, the gas is cooled and it turns back into a liquid (recondenses). The recondensed liquid is collected in a separate container. Eventually all of the parts of the liquid-liquid mixture will vaporize and then recondense in a separate chamber to be collected in separate containers
Processing Petroleum
•Petroleum is a naturally occurring mixture of different hydrocarbons.
• Holes are drilled into the ground to extract the raw petroleum, which we call “crude.” In this state, petroleum is unuseable. It must be refined in order to become useful.
• In different refining methods, mixture separation is occurring.
• Holes are drilled into the ground to extract the raw petroleum, which we call “crude.” In this state, petroleum is unuseable. It must be refined in order to become useful.
• In different refining methods, mixture separation is occurring.
•Fractional distillation: a liquid from liquid distillation process.
• Petroleum is a collection of substances that are soluble in each other but not in water. Each substance condenses at a different temperature. Fractional distillation is done in a two-tower structure.
• In the shorter tower, the petroleum is heated strongly enough to vaporize every part of the mixture. Then the mixture of hot vapours is pumped into the bottom of the taller tower.
• Petroleum is a collection of substances that are soluble in each other but not in water. Each substance condenses at a different temperature. Fractional distillation is done in a two-tower structure.
• In the shorter tower, the petroleum is heated strongly enough to vaporize every part of the mixture. Then the mixture of hot vapours is pumped into the bottom of the taller tower.
- Inside the tall tower, the hot vapours rise. As they rise, they cool. Remember that these are different pure substances, so they have different properties. This means that some of them condense and form a liquid while they are still very hot, near the bottom of the tower. As the remaining vapours continue to rise, different ones condense at different levels in the tower. Near the top (the coolest part) of the tower, a few remain as a gas.
• Each fraction is drawn off by the collecting pipes at its own level and is sent to a different part of the refinery for further processing.
Solid Mixtures
• Many valuable minerals are often mixed in with other solid materials.
•Think of when you break open a rock and you see all the different textures, colours etc.
•Ore: is a a mineral or group of minerals that contain a valuable substance (such as gold).
•Think of when you break open a rock and you see all the different textures, colours etc.
•Ore: is a a mineral or group of minerals that contain a valuable substance (such as gold).
• There are many different processes and procedures to separate ore:
1. Ore is blasted and then crushed
2. Ore powder is mixed with water to create a fine suspension
3. Chemicals are added to dissolve the gold
4. The mixture is run through a filter, where the dissolved gold goes through the filter, but the larger parts are filtered out.
5. Zinc is added to return the gold to its original state.
1. Ore is blasted and then crushed
2. Ore powder is mixed with water to create a fine suspension
3. Chemicals are added to dissolve the gold
4. The mixture is run through a filter, where the dissolved gold goes through the filter, but the larger parts are filtered out.
5. Zinc is added to return the gold to its original state.
Applications
1.Convenience foods are often dehydrated, so they can be stored for long periods of time without spoiling (you just add water )
2.petrochemical products include aspirin, sports equipment, eyeglasses, chewing gum, duct tape and fertilizer
3.there are many different types of salt, the most common one is sodium chloride (table salt) , potassium chloride is potash
4.sugar beets and sugar cane are refined to give us sugar crystals, maple sap is boiled to make ‘'sweetwater’' or maple syrup
2.petrochemical products include aspirin, sports equipment, eyeglasses, chewing gum, duct tape and fertilizer
3.there are many different types of salt, the most common one is sodium chloride (table salt) , potassium chloride is potash
4.sugar beets and sugar cane are refined to give us sugar crystals, maple sap is boiled to make ‘'sweetwater’' or maple syrup